Oxonium ions – molecules containing a formally positively charged oxygen atom with three chemical bonds – play an important role in chemistry. Meerwein’s salt is perhaps the best known example of a trialkyloxonium ion and is one of the post potent methylating reagents used by organic chemists. However, the closely related allylic, benzylic, propargylic, and allenylic oxonium ions have remained largely unexplored. Now, in a collaborative study involving Oxford Chemistry and recently published in Nature Synthesis, scientists have developed a method to prepare a range of new oxonium ions.
Allylic and benzylic oxonium ions have long been proposed as reactive intermediates. The only examples to be well-characterised so far have typically been in molecules that are rigid and highly constrained. More flexible oxonium ions have, until now, received little attention.
In a collaborative study between the synthetic group of Jonathan Burton at the University of Oxford and the computational group of Robert Paton at Colorado State University, chemists have reported a general and flexible method to prepare a range of these unusual oxonium ions that do not rely on highly constrained scaffolds. Using this approach, the team have successfully synthesised allylic and benzylic oxonium ions, along with previously unreported propargylic and allenylic variants.
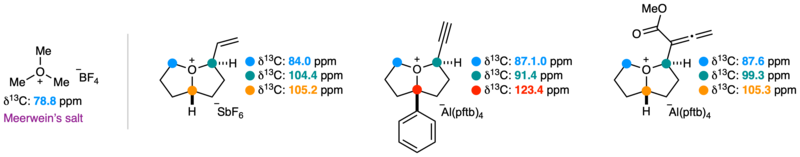
Carbon-13 NMR data for a selection of the synthesised oxonium ions.
Importantly, most of these oxonium ions were sufficiently stable to be characterised at room temperature by NMR spectroscopy. This was supported by structural and mechanistic analysis through density functional theory calculations. During the team’s investigation, they also discovered several unexpected reactions and distinctive behaviours of these spec ies, giving new insight into their reactivity.
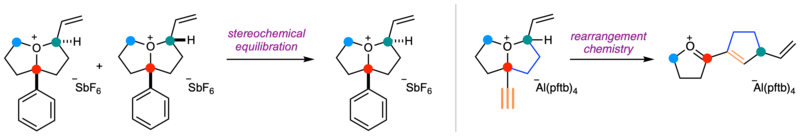
Equilibration and rearrangement chemistry of two of the synthesised oxonium ions.
Sam Chan, who led the synthetic work, commented:
It was certainly a daring attempt to go after unconstrained oxonium ions with allylic, benzylic, propargylic and allenylic substitutions. We were not entirely convinced the synthesis would be successful given the expected stability issues of these exotic ions. However, curiosity took the reins, and we gave it a shot anyway. I still vividly remember the day we got the proton NMR spectrum of the unconstrained allylic oxonium ion during teatime, and it was pristine. That gave us the clue to march forward and the rest is history.
Read more in Nature Synthesis.