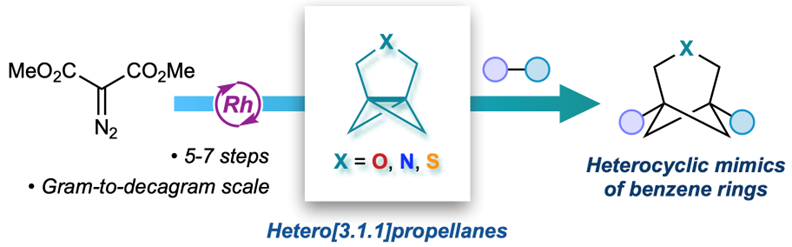
Propellanes are molecules containing a carbon–carbon bond to which three rings are fused. When two or more of these rings are cyclopropanes, the molecules are highly strained and readily undergo reactions that cleave their central carbon–carbon bond. The products of these ring-opening reactions are bridged bicycloalkanes, motifs that have recently emerged as surrogates for benzene rings in drug design which offer superior physical and chemical properties compared to their aromatic counterparts.
Despite the utility of these fascinating molecules, small ring propellanes containing heteroatoms such as oxygen, nitrogen or sulfur have to date eluded synthetic chemists. Now, researchers from Oxford have developed a 'unified' strategy to prepare three of these 'heterocyclic propellanes' and thereby open up a direct route to heteroatom-containing bicycloalkanes which are similarly of high interest to medicinal chemists. This research, led by Prof Ed Anderson and DPhil student Rebecca Revie with the support of Dr Ayan Dasgupta, has been published today in Nature Chemistry.
A scalable route to new heterocyclic small ring propellanes, precursors to modern mimics of benzene rings for drug design
Key to the success of this project was the development of a robust route to the propellane precursors, which required an entirely new synthetic approach compared to their established carbocyclic analogues. Rebecca developed routes that could be conducted on multigram or even decagram scale, with the propellanes accessed in just 5–7 synthetic steps. Carbocyclic propellanes are notoriously unstable at room temperature, cannot be isolated neat due to spontaneous polymerisation, and must be purified by distillation. However, the team found these new heterocyclic propellanes are stable for several days on the benchtop, did not require the anticipated distillation, and in the case of nitrogen-containing propellanes could even be isolated in crystalline form.
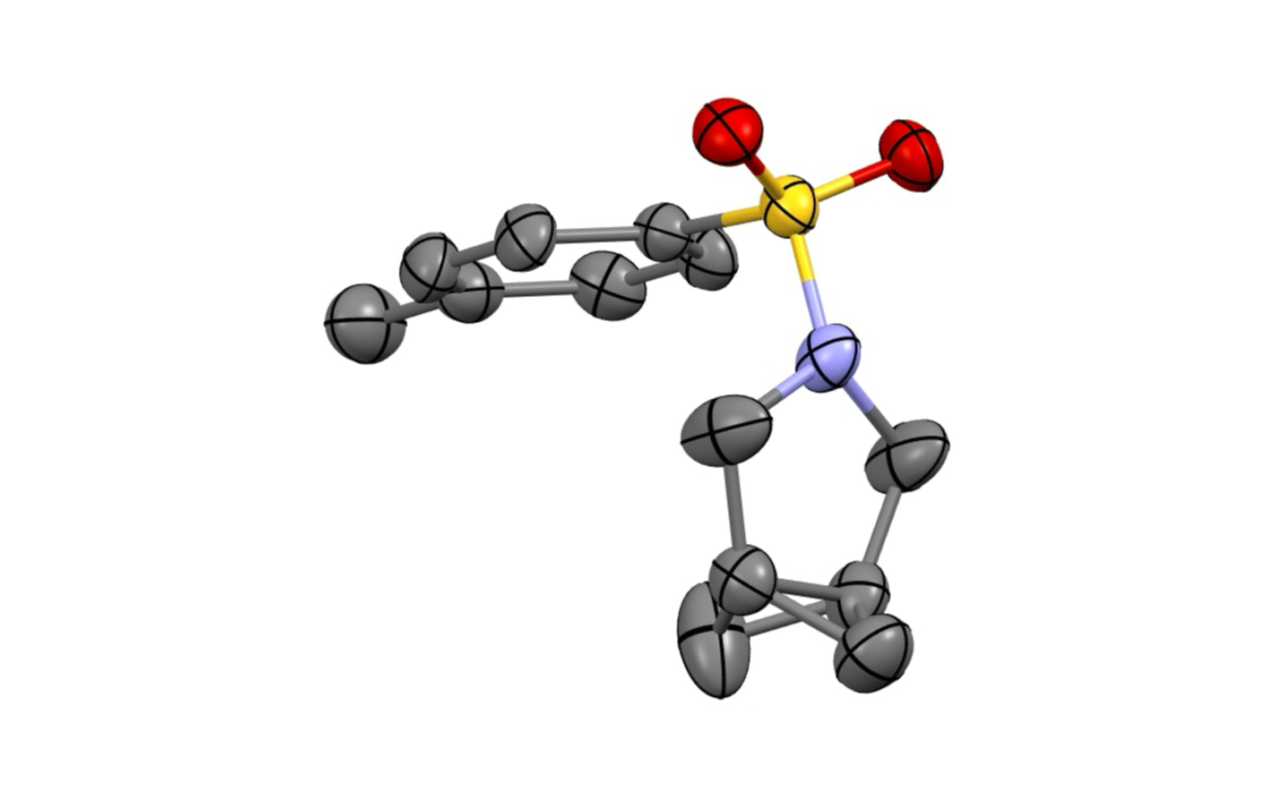
The X-ray diffraction crystal structure of an aza-propellane
A further key aspect of this work was collaboration with the pharmaceutical company AbbVie, and specifically process chemist Dr Russell Smith. As well as funding the project, AbbVie chemists were able to evaluate safety aspects of these strained molecules, and explore some properties of the ring-opening products. Prof Anderson commented: "This research paves the way to a new generation of propellane molecules, and also offers a valuable and direct route to building blocks that are highly sought after in the pharma industry. We're excited to explore the properties and reactivity of these unique compounds, and to push the frontiers of propellane research even further."
Read more in Nature Chemistry.