Controlling cell membrane transport with light
How can interactions between molecules be controlled when they are confined within a cell membrane? This highly biologically relevant question is one of those being considered by Matthew Langton’s research group, established in the Chemistry Department in 2018.
All biological cells are separated from their environment by the cell membrane, a wall-like structure made up of two layers of fatty molecules. This structure, known as a lipid bilayer, and the proteins embedded within it, regulate what can enter or exit the cell’s interior.
Understanding and controlling the flow of molecules or ions across this membrane may help in developing new treatments for diseases such as cystic fibrosis, whereby a protein that controls the flow of chloride ions across the cell membrane is not functioning correctly in the body. As well as its relevance to pure biological research, supramolecular chemistry in cell membranes has applications in catalysis.
If a catalyst can be embedded into a membrane then reactions could be performed in an artificial cell and controlled by chemical signalling – for a review of supramolecular chemistry in bilayer membranes see the references below.
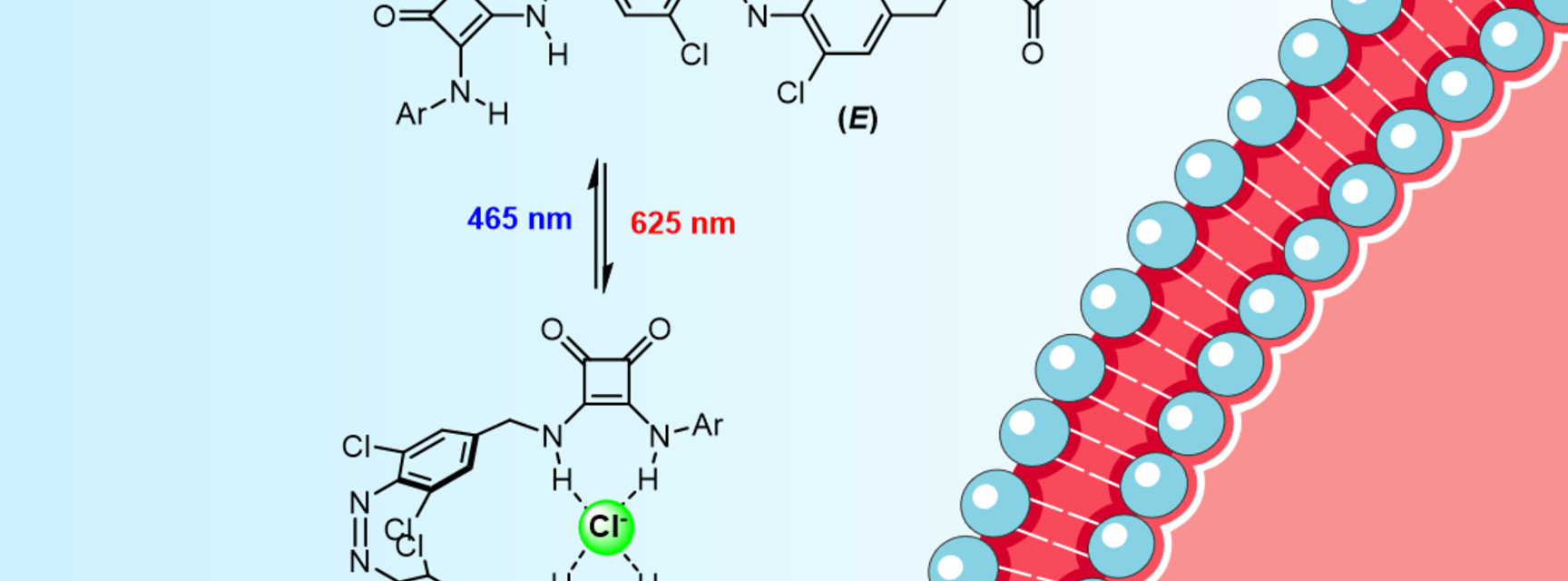
Transport across cell membranes in biology is controlled by proteins that respond to a stimulus, be it chemical or physical. In a recent study, Langton and DPhil student Aidan Kerckhoffs designed, synthesized, and characterised an artificial chloride ion transporter that can be controlled using visible light. A specially designed linear molecule undergoes a reversible isomerization under red light to a curved form, which can bind very effectively to a single chloride ion using hydrogen bonds (see figure). The curved form can then be transformed back into the less efficiently binding linear form using blue light.
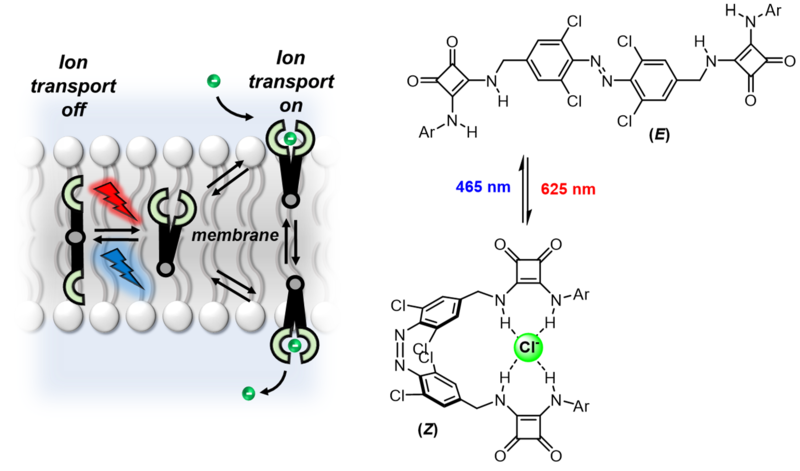
A specially designed artificial chloride ion transporter undergoes an isomerization between linear and curved forms under red light, which is reversed by blue light. The curved form transports chloride ions across the lipid bilayer much more effectively than the linear form, meaning light can be used to control the rate of chloride ion flow across the membrane.
It was recently demonstrated that when the transporter molecule is incorporated into a lipid bilayer, the reversible photoswitching can be used to reliably accelerate and decelerate the rate of chloride ion transport across the membrane. As well as increasing the rate with red light, it can also be slowed back down by applying blue light, which forces the molecule back into its linear form. Previous studies have used ultraviolet (UV) light to activate ion transporters, but not in this reversible way. Visible light also presents advantages over UV for potential therapeutic applications because it can penetrate into the tissue more effectively and does not cause collateral damage to cells.
“Supramolecular chemistry in general is very collaborative,” says Matthew Langton, “it stretches from material science and molecular machines through to chemical biology and everything in-between”. Ultimately, research of this kind may lead to the use of cell-like compartments as small reaction vessels, by controlling how reactants, catalysts, and products enter and exit the interior of the cell, opening up a world of biologically inspired reactions that can be performed and controlled on the nanoscale.
References:
Cell membrane diagram in banner image from Servier Medical Art.