New frontiers in radiochemistry with an unexplored 18F-labelled carbene
New frontiers in radiochemistry with an unexplored 18F-labelled carbene
Researchers from Professor Véronique Gouverneur’s group in Oxford Chemistry, working in collaboration with Cardiff University, Colorado State University, and industrial partners from Pfizer, Janssen and UCB, have developed a novel method to generate fluorine-based chemical tracers which will aid in disease diagnosis and drug discovery.
The results of their study, which are published today in Nature, unveil for the first time access to fluorine-18 (18F) labelled difluorocarbene, a powerful new tool capable of generating a wide variety of biologically and medicinally relevant molecules.
18F is an artificially generated radioactive isotope that emits positrons, a subatomic particle that, upon annihilation with an electron, releases gamma rays that can be used to image the body in a similar way to an X-ray scanner. Unlike X-ray imaging, which requires an external source, positron emission tomography (PET) can track the movement of molecules that contain a radioisotope such as 18F throughout the body and use it to monitor metabolic processes related to disease or provide information on a drug molecule’s behaviour in the body.
18F has previously been successfully incorporated into molecules where fluorine is present either on an aromatic ring or as a single fluoride group. Many candidate drug molecules, however, contain difluoromethyl groups, denoted as CF2H. These can’t be radiolabelled efficiently with 18F using established synthesis methods. To make the CF2H motif it is more common to use a more reactive species known as a difluorocarbene (DFC), which in its 18F-radiolabelled form has, until now, never been successfully used in radiosynthesis.
Using an overlooked reaction from the literature, the Oxford team hit upon a reagent that appeared to generate a carbene and could be formed using [18F]fluoride ions. From preliminary experiments they knew the reagent was accessible in 18F-labelled form. The real challenge was to establish whether this reagent could release [18F]difluorocarbene and, more importantly, whether they could harness its reactivity to access a wide array of CF2H radiolabelled products in high molar activity (a measure of the amount of product formed that contains the radioactive 18F isotope). High molar activity radiotracers are essential to successfully image many disease states, and access to radiolabelled molecules containing CF2H groups in high molar activity remains a holy grail challenge. Exploiting the power of 18F-labelled difluorocarbene brings us one step closer to achieving this goal.
Dr Jeroen Sap, a postdoctoral researcher in the Gouverneur group and one of the lead authors of the study, says:
We successfully tackled the molar activity problem by employing physical organic chemistry experiments, deciphering the key contributing factors affecting molar activity and redesigning the DFC reagent to meet this challenge. We can now take a well-explored reactive intermediate routinely used in organic synthesis and exploit its 18F version to significantly widen the landscape of PET radiotracers accessible for imaging applications.
The researchers went on to demonstrate that this 18F-labelled DFC can be used to successfully synthesize dozens of common CF2H-containing derivatives of known tracer or drug molecules, and further that these molecules can be generated quickly and used as PET imaging agents.
18F has a half-life of 110 minutes, meaning that it decays into stable isotopes in a matter of hours. Although this makes it safe to use in humans, it also limits the time window for PET imaging, with a race against the clock meaning that any chemical synthesis involving freshly generated 18F must happen in a matter of hours. To fast track the synthesis, the team automated the production of the radiotracer using a computer-controlled synthesis robot (TRASIS AllinOne), in collaboration with Cardiff University. This automated protocol will help the broader research community to adopt their method.
Once the carbene they generated had been used to label a radiotracer that targets a protein commonly associated with neurodegenerative diseases, they tested it in a mouse-model for Huntington’s disease. Through PET imaging studies they illustrated that their radiotracer could be used to image the mouse’s brain and that it was preferentially taken up in particular areas of the brain associated with disease.
Professor Gouverneur says:
Expanding the radiochemical space available for the invention of the perfect PET radiotracer is the dream of all imaging scientists. We are now one step closer, having designed a reagent that releases a reactive 18F-labelled difluorocarbene capable of unlocking four new reactions for radiochemistry. We are very much looking forward to seeing how this novel radiochemistry will be deployed by the imaging community.
Read more about the study in Nature.
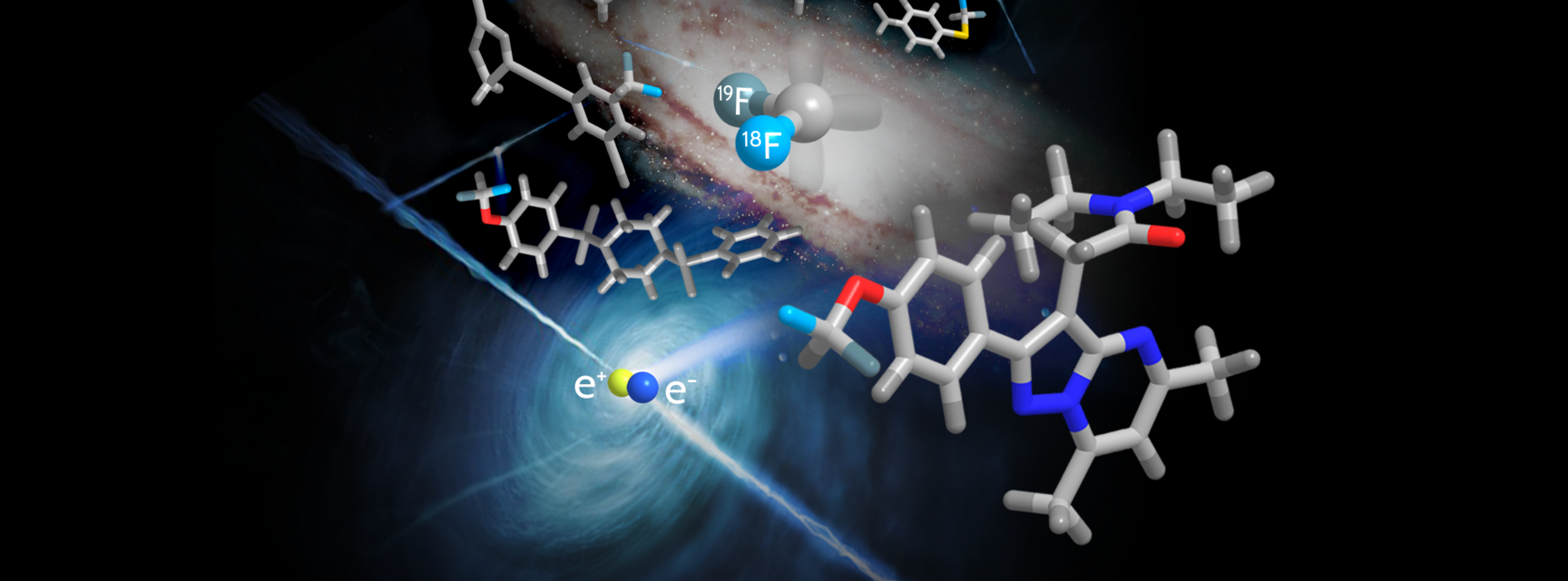
Illustration shows 18F-labelled DFC, molecules containing the CF2H group that the team synthesised using their DFC reagent, and a representation of the positron annihilation event that is behind positron emission tomography. Illustration uses images courtesy of the ESO and Tengyart on Unsplash.