Spectroscopy to diagnose lung disease
Each time you breathe in and out your lungs process a mixture of gases: oxygen is consumed to fuel the body, while carbon dioxide and water are produced. Tracking the levels of these gases as a patient breathes would allow clinicians to accurately assess how efficiently the lungs are functioning, which is important when diagnosing and treating diseases from asthma and chronic obstructive pulmonary disease (COPD) to COVID-19.

The laser gas analyser in use, demonstrated by Dr Nick Smith, one of the chemists who developed it. The device is housed in the black box, which is breathed into by the subject using a mouthpiece and noseclip to ensure that all inhaled and exhaled gases enter the device.
The Ritchie group have been collaborating with colleagues in the Department of Physiology, Anatomy, and Genetics to develop a laser gas analyser, which non-invasively measures the concentrations of these gases at the mouth.
Using laser absorption spectroscopy, the analyser records the concentrations of O2, H2O, and CO2 100 times a second with an unprecedented level of accuracy while a patient sits and breathes into the device. By also measuring the temperature and pressure, the perfect gas law can be used to infer the amount of nitrogen, since these four gases make up over 99% of the air around us.
Direct absorption spectroscopy, probing vibrational transitions in the near-infrared spectral region, is used to detect the H2O and CO2. For O2 the more sensitive technique of cavity-enhanced absorption spectroscopy (CEAS) is used to probe an electronic transition. In CEAS the laser light is trapped between a pair of mirrors and passes through the sample many times before being detected, greatly increasing the sensitivity.
The gas concentrations are combined with flow measurements to generate highly precise profiles of gas being taken in or produced by the lungs. Using a custom mechanistic model of the lung it is possible to calculate clinically valuable parameters such as the variation in the ventilation and blood supply of gas exchange compartments within the lung. A healthy lung should show a narrow distribution of these quantities, since the alveoli within the lungs should all work relatively well, whereas a lung with reduced function will show a wider variation, indicating that many of the alveoli have deteriorated.
It has been demonstrated that this gas analyser can be used to measure variation in lung function between groups of young, old, and COPD-diagnosed individuals. Differences in clinically relevant parameters were found between all three groups, with significantly wider distributions for the patients with COPD. A further study has also shown that the laser gas analyser can provide measures that are more sensitive than current clinical tools at assessing deterioration in lung function associated with badly controlled asthma. Faster diagnosis and more accurate assessments of these respiratory diseases would allow more effective interventions and, the hope is, better clinical outcomes.
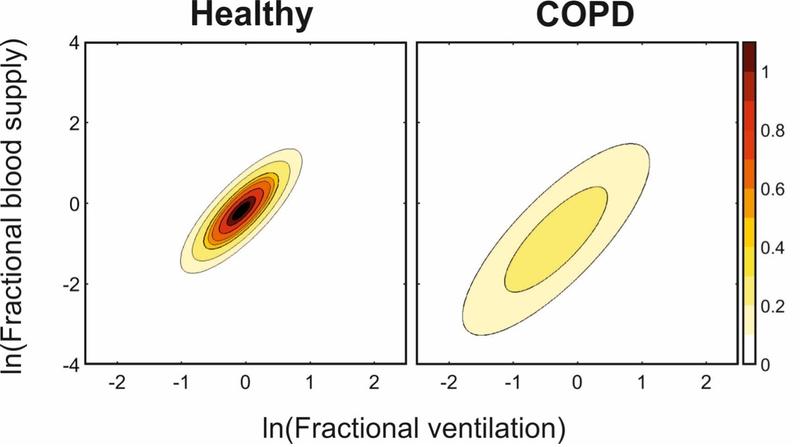
The distributions of both blood supply (vertical axis) and ventilation (horizontal axis) across the lung are found to be significantly wider for COPD patients (right) than for young healthy individuals (left).
This newly developed technique is currently being used to assess the lung function of patients who have been hospitalized with COVID-19, testing the hypothesis that the long-term effects of the disease, sometimes known as “long COVID”, may involve a breakdown in the lung’s ability to divert blood away from damaged regions.
References:
Banner photo of a lung model by Robina Weermeijer on Unsplash