Chemistry Teaching Laboratory Workshops
General Information
The undergraduate teaching laboratories at the University of Oxford are world-class, providing unparalleled facilities for our students. Where possible, outside of term-time these laboratories are made available to state school teachers and young people studying GCSE and A level Chemistry (or equivalent) to support their study of the chemical sciences. You can find details of available workshops and activities on our 'What's On?' page.
Please note that the construction works of the Life and Mind Building is curtailing access to the CTL outside of University terms.
Location
The new Chemistry Teaching Laboratories (Tel: 01865 281505) are located just off South Parks Road. However, due to current building works, the CTL is accessed via Mansfield Road, using the postcode OX1 3SR.
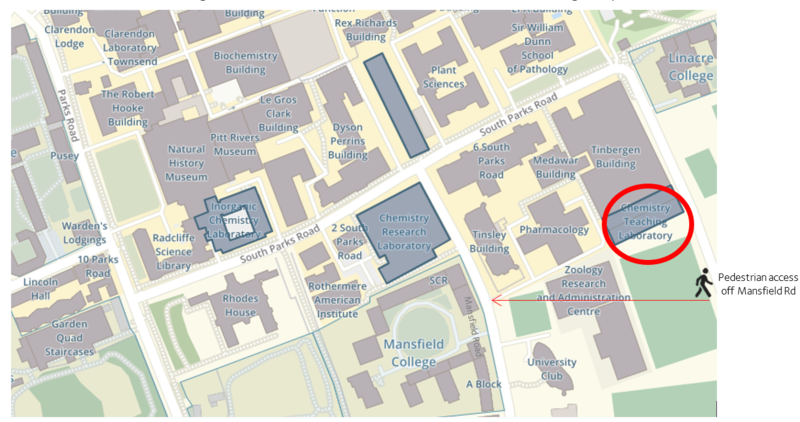
Regrettably, we are unable to provide parking for visiting schools or students. Please see ‘How to get to Oxford’ for further information on travelling to Oxford.
Catering
Food is not provided for visiting groups (unless otherwise stated in the event information); you will need to bring your own packed lunches/snacks. There is seating and tables in the CTL foyer, a drinking water fountain, as well as a vending machine that can be used by groups booked to visit the CTL for a workshop / organised activity.
Bag storage
Lockers are provided for use by visiting groups. A limited number of padlocks are available from reception for teachers wishing to store valuables.
Photographs and social media
Visiting teaching staff have found that images taken in the laboratory make good copy for school websites, newsletters and social media and you are welcome to take photographs in the CTL. The University of Oxford is keen to promote its outreach activities through the use of photographs taken with visiting groups. Please read and sign the accompanying consent form found in our Health and Safety tab.
Health and Safety
Visitors to the CTL are required to read and abide by the Health and Safety information for visitors and visiting groups. Please ensure you read the Health and Safety information on this site, and download and sign the accompanying consent form. Please note that the CTL staff reserve the right to refuse entry to any student or staff who is not dressed appropriately for working in a laboratory.
Risk Assessments
Risk assessments are available for all of our workshops and will be sent out on confirmation of a workshop booking. Please contact outreach@chem.ox.ac.uk if you require an additional copy.
A pdf copy of the information above can be downloaded here.
Health and Safety
Visitors to the CTL are required to read and abide by the Health and Safety information for visitors and visiting groups, below.
If you are visiting as a school group, please download and sign the group consent form and the data sharing agreement form.
If you are visiting as a solo student, please download and sign the solo consent form. Please note, we only accept bookings from solo students if you are unable to be accompanied by their teacher. Presently, there is a limit of 6 solo students from any one school. You must be over 16 yrs old on the day of your workshop.
Your data will be processed according to our privacy policy.
Please note that the CTL staff reserve the right to refuse entry to any student or staff who is not dressed appropriately for working in a laboratory.
General Safety
Sensible, acceptable behaviour is expected at all times and is the responsibility of the teacher in charge of the visit. Visiting students who fail to follow basic safety rules may be asked to leave the laboratory.
Sensible, acceptable behaviour is expected at all times and is the responsibility of the teacher in charge of the visit. Visiting students who fail to follow basic safety rules may be asked to leave the laboratory.
Acceptable Clothing for Laboratory Work
To ensure safety in the laboratory, all individuals must wear appropriate clothing. The Department reserves the right to refuse entry to anyone who does not comply with these guidelines.
To ensure safety in the laboratory, all individuals must wear appropriate clothing. The Department reserves the right to refuse entry to anyone who does not comply with these guidelines.
- Clothing must fully cover the torso, including the chest, abdomen, back, pelvis, shoulders, and legs.
- Acceptable clothing includes short-sleeved t-shirts, long-sleeved tops, and trousers made of sturdy material that provides adequate protection.
- School uniforms are only acceptable if they adhere to these requirements.
Prohibited Clothing:
- No bare skin should be visible once a lab coat is worn, except for the hands and face.
- Crop tops, strappy tops, and tops with open backs are not permitted, as they expose vital areas of the body.
- Sandals, open-toe shoes (e.g., ballerina pumps), and high heels are strictly prohibited.
- Lycra-style sports leggings, tights, and thin-material trousers are not allowed, as they do not offer sufficient protection against chemical spills or splashes.
- Shorts, short skirts, cropped trousers, ripped jeans, and trainer socks (ankles must be covered) are not permitted.
- Additionally, long hair must be securely tied back.
Safety glasses and laboratory coats will be provided by the Oxford University Chemistry Department.
Additional Lab Rules
• No food or drink is to be consumed (including gum and medication) in the labs.
• No cosmetics or skin preparations may be applied.
• Hands must be washed after leaving the laboratory.
• No food or drink is to be consumed (including gum and medication) in the labs.
• No cosmetics or skin preparations may be applied.
• Hands must be washed after leaving the laboratory.
Special Requirements
To prepare our demonstrators effectively for the visit please inform us of any specific needs of students in your group. This includes specific physical requirements or special educational needs.
We regret that crutches are not permitted in the laboratory as they cause a trip hazard.
We have accessible spaces for wheelchair users, which can be prepared with advance warning.
To prepare our demonstrators effectively for the visit please inform us of any specific needs of students in your group. This includes specific physical requirements or special educational needs.
We regret that crutches are not permitted in the laboratory as they cause a trip hazard.
We have accessible spaces for wheelchair users, which can be prepared with advance warning.
Risk Assessments
Risk assessments are available for all of our workshops and will be sent out on confirmation of a workshop booking. Please contact outreach@chem.ox.ac.uk if you require an additional copy.
HEAT Database
Visiting students are asked to submit their details to our HEAT database. The information the University of Oxford collects and records in HEAT means that we can assess whether the outreach activities we fund and deliver make a difference to the education and career choices that young people make. This information will never be used by the University of Oxford to contact you for marketing any kind of product, service or university course, and we will never allow anybody else to use it for this purpose.






