Kylie Vincent

Professor Kylie Vincent
Professor of Inorganic Chemistry
Associate Head of Department (People)
The Vincent group are interested in both understanding and applying catalysis at metal sites within enzymes.
We are particularly interested in small molecule activation at metal sites in enzymes. Chemistry has a crucial role to play in solving the pressing global challenges of sustainable energy supply, and capture and use of carbon dioxide. In these areas, nature has already established exquisitely tuned enzymes as catalysts. Redox enzymes play key roles as catalysts in biological cycling of hydrogen, carbon and nitrogen and energy. Their active sites are built from common metals such as iron, nickel and copper, and the reactivity achieved at these biological metal centres is often difficult to replicate under ambient conditions with synthetic catalysts. We are interested in understanding details of the redox chemistry and mechanisms of catalysis occurring at enzyme active sites. A central theme in our research is to bridge the gaps between different techniques in bioinorganic chemistry. We combine electrochemistry and spectroscopy to control and study enzymes as they engage in catalytic turnover. We apply these approaches to single crystals of enzymes to bridge the gaps between solution and solid state measurements and reveal structures of catalytically active intermediates. Our studies target a range of metalloproteins including hydrogenases, nitrogenase, carbon monoxide dehydrogenase and nitric oxide sensing proteins. We are particularly interested in the efficient catalysis of H2 oxidation and production by microbial hydrogenase enzymes at [NiFe] active centres. Ligands at the active site of hydrogenases, CO and CN-, give rise to fairly intense absorption bands in the Infrared spectrum. This has inspired us to develop new approaches to infrared spectroelectrochemistry for studying hydrogenase active site chemistry under direct electrochemical control. Understanding and exploiting selective nature's catalysts should provide inspiration for development of new classes of energy-cycling catalysts.
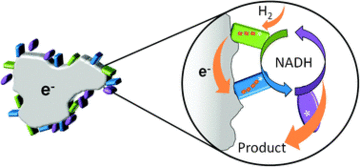
We are also fascinated by applications of enzymes in chemical synthesis. We have developed and patented the HydRegen system for C=X bond hydrogenations via H2-driven recycling of the biological hydride transfer cofactors, NADH and NADPH. This offers the best of both worlds between the impressive selectivity of biocatalysis on the one hand, and the atom-efficiency and ease of implementation of hydrogenations (which are typically catalysed by precious metals). We have demonstrated this approach for a varienty of C=X bond reductions, using a wide range of NAD(P)H dependent enzymes, including keto reductases, imine reductases, ene reductases and amino acid and amine dehydrogenases. We are working closely in collaboration with industry to develop the HydRegen system for industrial fine chemical synthesis. In recent work, we have extended this approach to selective biocatalytic deuterium insertion, and operated many of our biocatalytic reactions in continuous flow.
Kylie Vincent's research is supported by the EPSRC, BBSRC and InnovateUK, and the European Research Council. We are also supported by collaboration with a variety of industrial partners including Johnson Matthey and Dr Reddy's.
Kylie Vincent is a graduate of the University of Melbourne, Australia (BA/ BSc(Hons), Ph.D. (2004)). Her PhD research was conducted in the group of Stephen Best (University of Melbourne) and involved a 12 month collaborative visit to the group of Chris Pickett (John Innes Centre, Norwich, UK) supported by a Rae and Edith Bennett Travelling Scholarship. She carried out postdoctoral research at the University of Oxford with Fraser Armstrong from 2002-2007 and held an R. J. P. Williams Junior Research Fellowship at Wadham College, Oxford from 2003-2007.
In October 2007 she took up a Royal Society University Research Fellowship in the Chemistry Department at Oxford University and a Senior Research Fellowship at Wadham College, Oxford. In October 2008 she was appointed to an RCUK Academic Fellowship in the Chemistry Department and a Senior Research Fellowship at Jesus College Oxford. She was appointed Associate Professor in 2013, and appointed to a full Professorship in 2017.
Her research interests include mechanistic studies of small molecule activation in biology, and applications of enzymes in chemical synthesis. She teaches in Inorganic Chemistry.




