New ‘droplet battery’ could pave the way for miniature bio-integrated devices
New ‘droplet battery’ could pave the way for miniature bio-integrated devices
- Researchers have developed a miniature battery that could be used to power tiny devices integrated into human tissues.
- The design uses an ionic gradient across a chain of droplets – inspired by how electric eels generate electricity.
- The device was able to regulate the biological activity of human neurons.
- This could open the way to the development of tiny bio-integrated devices, with a range of applications in biology and medicine.
University of Oxford researchers have made a significant step towards realising miniature bio-integrated devices, capable of directly stimulating cells. The work has been published today in the journal Nature.

Bright-field images of a droplet power source. Left: Enlarged version of the droplet power source, for visualisation. 500 nL volume droplets were encapsulated in a flexible and compressible organogel. Scale bar: 10 mm. Right: Zoom in view of an actual-sized droplet power source, made of 50 nL droplets. Scale bar: 500 μm. Image credit: Yujia Zhang.
Small bio-integrated devices that can interact with and stimulate cells could have important therapeutic applications, including the delivery of targeted drug therapies and the acceleration of wound healing. However, such devices all need a power source to operate. To date, there has been no efficient means to provide power at the microscale level.
To address this, researchers from the University of Oxford’s Department of Chemistry have developed a miniature power source capable of altering the activity of cultured human nerve cells. Inspired by how electric eels generate electricity, the device uses internal ion gradients to generate energy.
The miniaturized soft power source is produced by depositing a chain of five nanolitre-sized droplets of a conductive hydrogel (a 3D network of polymer chains containing a large quantity of absorbed water). Each droplet has a different composition so that a salt concentration gradient is created across the chain. The droplets are separated from their neighbours by lipid bilayers, which provide mechanical support while preventing ions from flowing between the droplets.
The power source is turned on by cooling the structure to 4°C and changing the surrounding medium: this disrupts the lipid bilayers and causes the droplets to form a continuous hydrogel. This allows the ions to move through the conductive hydrogel, from the high-salt droplets at the two ends to the low-salt droplet in the middle. By connecting the end droplets to electrodes, the energy released from the ion gradients is transformed into electricity, enabling the hydrogel structure to act as a power source for external components.
In the study, the activated droplet power source produced a current which persisted for over 30 minutes. The maximum output power of a unit made of 50 nanolitre droplets was around 65 nanowatts (nW). The devices produced a similar amount of current after being stored for 36 hours.
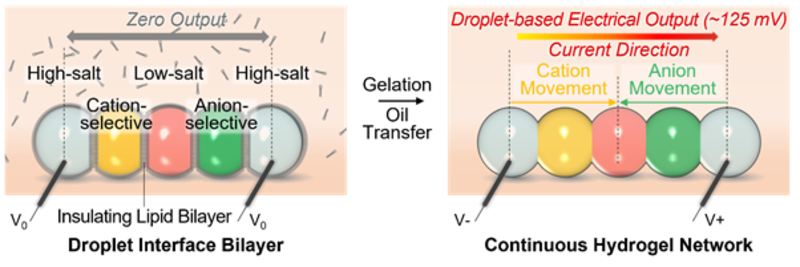
The activation process for the hydrogel droplet power unit. Left, before the battery is activated, an insulating lipid prevents ion flux between the droplets. Right: The power source is activated by a thermal gelation process to rupture the lipid bilayers. Ions then move through the conductive hydrogel, from the high-salt droplets at two ends to the middle low-salt droplet. Silver/silver chloride electrodes were used to measure electrical output. Image credit: Yujia Zhang.
The research team then demonstrated how living cells could be attached to one end of the device so that their activity could be directly regulated by the ionic current. The team attached the device to droplets containing human neural progenitor cells, which had been stained with a fluorescent dye to indicate their activity. When the power source was turned on, time-lapse recording demonstrated waves of intercellular calcium signalling* in the neurons, induced by the local ionic current.
Dr Yujia Zhang (Department of Chemistry, University of Oxford), the lead researcher for the study, said:
The miniaturized soft power source represents a breakthrough in bio-integrated devices. By harnessing ion gradients, we have developed a miniature, biocompatible system for regulating cells and tissues on the microscale, which opens up a wide range of potential applications in biology and medicine.
According to the researchers, the device’s modular design would allow multiple units to be combined in order to increase the voltage and/or current generated. This could open the door to powering next-generation wearable devices, bio-hybrid interfaces, implants, synthetic tissues, and microrobots. By combining 20 five-droplet units in series, they were able to illuminate a light-emitting diode, which requires about 2 Volts. They envisage that automating the production of the devices, for instance by using a droplet printer, could produce droplet networks composed of thousands of power units.
Professor Hagan Bayley (Department of Chemistry, University of Oxford), the research group leader for the study, said:
This work addresses the important question of how stimulation produced by soft, biocompatible devices can be coupled with living cells. The potential impact on devices including bio-hybrid interfaces, implants, and microrobots is substantial.
* Calcium signalling is a key mechanism through which neurons communicate to one another to coordinate biological activities such as neurotransmitter release, neuronal firing, synaptic plasticity, and gene transcription.




